Prime Scientific - Theory and Practice of Spectrophotometric Measurements
In this application note, we discuss the theory behind the measurements based on spectrophotometry
·
2 min read

This law shows that absorbance is linear to concentration but this is only true for low concentrations. For
absorbance levels above 3 the concentration starts to move away from the linear relationship.
Transmittance is the proportion of the light which passes through the sample:
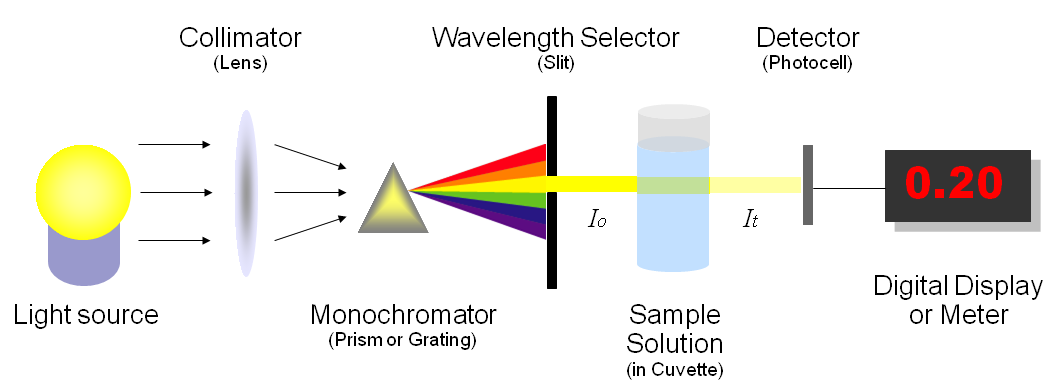
Spectroscopy Measurement
There are four main components of a spectrophotometer. These are a light source to emit a high and
constant amount of energy over the full wavelength range; a method for separating the light into
discreet wavelengths; a sample holder and a light detector.
The light from the pre-focused tungsten halogen (7310) or pre-aligned xenon (7315) lamp is focused
onto the grating, with 1200 lines per millimeter, which separates the light into discreet wavelengths.
The diffracted spectrum of light then passes through a further slit and lens arrangement before passing
through the sample in the sample chamber from left to right. The light which is not absorbed by the
sample is transmitted through a collecting lens and onto the signal detector. The photo-diode detector
used is mounted directly onto the detector PCB and the output is used to calculate the % transmittance.
The result is displayed either as % transmittance or absorbance on the instrument display.
For more information on our spectrophotometers range, please visit here
In our upcoming blogs, we will discuss the best practice guidelines of properly using your spectrophotometers and the most probable causes of issues of operation.
References:
Prime Scientific
Prime Scientific provides equipment, instruments and other related products and materials. We aim to be a one stop shop for all our customers laboratory needs. Since 2001, Prime has strived to provide the best services and solutions to the scientific community in the region.